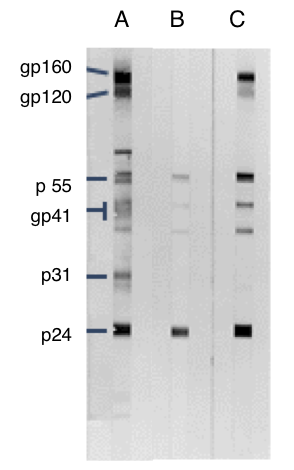
Most commercially available EIAs have a high sensitivity and specificity and are able to detect all subtypes of HIV-1 and HIV-2. Infants born to HIV-infected women may therefore initially test seropositive, irrespective of their own infection status. However, because maternal HIV antibodies (immunoglobulin G ) are passively transferred across the placenta, HIV serological assays in infants are difficult to interpret. Antibodies to HIV-1 and HIV-2 are detected by EIA, also known as enzyme-linked immunosorbent assay (ELISA), simple/rapid test devices, and western blot (WB) tests. 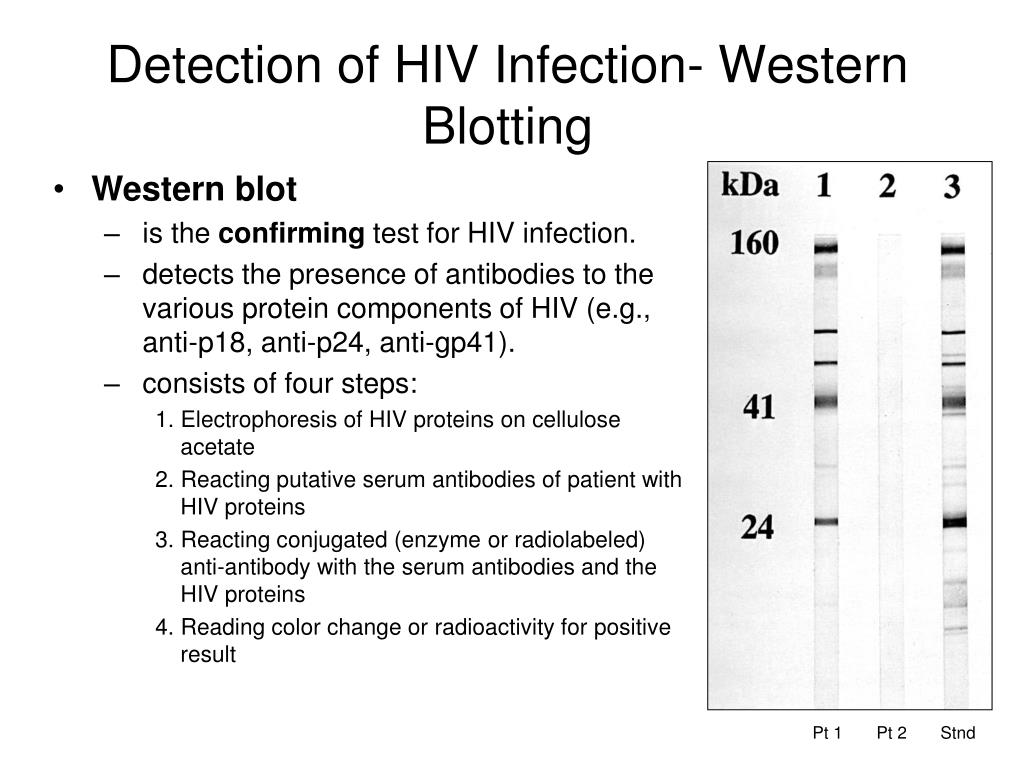
None of these detect HIV itself, but rather detect an immune response to the virus, and therefore take some time to develop and become reactive (or positive) after HIV infection has been acquired. It is recommended that countries use serological assays (rapid tests or EIAs) within a testing algorithm that has been validated in-country by the National Reference Laboratory (or another laboratory designated for this purpose).Īntibodies to HIV can be measured by a variety of techniques. WHO Prequalification is a voluntary process, usually at the request of the manufacturer, and the results of these evaluations are made available online ( 32). Prequalification confers an eligibility to tender with United Nations (UN) agencies and enables bulk purchase. WHO, through the WHO Prequalification of Diagnostics Programme, evaluates the performance of commercially available technologies including serological assays (EIAs and rapid tests), CD4 enumeration technologies and molecular technologies. The sensitivity and specificity of different assays will vary, and test devices or equipment can be made by one company, but distributed and sold under several brand names. Non-commercial/in-house assays are not recommended for widespread use by national programmes, but may be used where ongoing and functional laboratory QA systems are in place and production is standardized. This is especially the case for in-house assays that lack the stringent quality control processes and technical support from the manufacturer that are available to commercial assays. Most of the assays utilized to detect nucleic acids (NAT) are complex, technically demanding or inappropriate for non-specialist diagnostic laboratories. Many methods for the detection of nucleic acids are available both commercially and non-commercially (in-house assays). Commercially versus non-commercially available tests Well-defined standard operating procedures (SOPs), following nationally defined and validated testing algorithms, are essential for optimal use of all laboratory-based testing.ĥ.2. Clinicians and staff providing laboratory services need regular communication about the performance of tests to improve and ensure appropriate performance. Therefore, ongoing QA within the context of the laboratory quality system, both internally and externally, is essential. 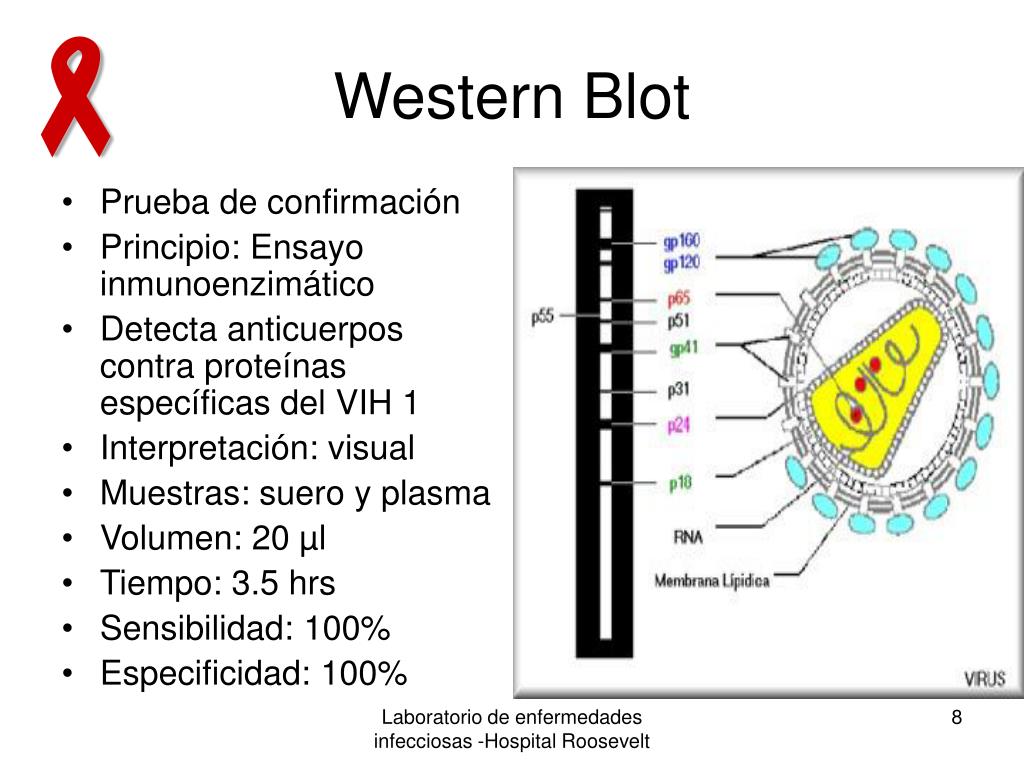
Many processes are involved from the time the specimen is collected and transported to the laboratory, tested and until the results are reported, during which errors can occur. 
Having highly accurate tests does not necessarily guarantee reliable laboratory results. In order to build laboratory capacity, national HIV/AIDS programmes should invest in a QA programme for all laboratories performing diagnostic testing, and should use existing available services provided by WHO and others including the Centers for Disease Control and Prevention (CDC) to support external quality assessment schemes (EQAS). A range of laboratory-based techniques are available, and these are discussed in more detail in the following section. In order to diagnose HIV infection definitely in children aged less than 18 months, assays are required that detect the virus or its components (i.e. Infected infants then go on to produce HIV antibody ( 30) however, most commonly used HIV serological assays cannot distinguish between maternal HIV antibody and HIV antibody produced by the infant, making the interpretation of reactive HIV serological test results difficult ( 29, 31). However, maternal HIV antibody is transferred to the baby passively during pregnancy and then declines (with a half-life of 28–30 days in non-breastfed infants) ( 24- 29). In children older than 18 months of age, serological testing should be used in the same manner as in adults. Serological testing identifies HIV antigen and/or antibody generated as part of the immune response to infection with HIV. The definitive diagnosis of HIV infection at any age requires diagnostic testing that confirms the presence of HIV.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
